Preliminary Call Deadline: 19 April 2026
PhD Topic 1: Solid-phase speciation and mobility of trace metal(loid)s (Cd, In, Ga, Ge) in sulfidic mine wastes

This project will investigate how the solid-phase speciation of trace metal(loid)s controls their mobility and stability in sulfidic mine wastes. The research will focus on the identification of host secondary minerals and the processes governing element release during oxidation of primary sulfides. The study will combine fieldwork at historical polymetallic mining sites (Czech Republic, Greece, Portugal) with advanced mineralogical and geochemical methods, including microanalysis and spectroscopy. The goal is to improve understanding of the environmental behavior and resource potential of critical metal(loid)s.
Supervisor: Prof. Petr Drahota (petr.drahota@natur.cuni.cz)
——————————————————————————————————————–
Topic 2: Behavior of used photovoltaic panels in an exogenous environment

Crystalline silicon solar panels are the most widely used type of photovoltaics worldwide. Their degradation can lead to release of toxic trace elements, making them emerging contaminants. The proposed topic of PhD will study weathering of used photovoltaic panels in soils and the release of toxic and critical elements. It includes both laboratory and in-situ experiments under different geochemical and environmental conditions (e.g., soil type, pH, redox conditions). The work involves geochemical analyses and isotope approaches to understand elements mobility, transformation, and pathways in the soil environment.
Supervisor: Dr. Maria Vaňková (maria.vankova@natur.cuni.cz)
Co-Advisor: Prof. Martin Mihaljevič
——————————————————————————————————————–
Workplace: Institute of Geochemistry, Mineralogy and Mineral Resources – Faculty of Science – Charles University – Prague – Czech Republic
Full-time PhD position
Starting date: 1 October 2026
Duration: 4 years
Salary conditions: doctoral scholarship + salary from a grant project
Required education: MSc degree
Required language skills: English (B2 Independent User) or Czech
——————————————————————————————————————–
How to apply: To apply, send an email to supervisors (petr.drahota@natur.cuni.cz or maria.vankova@natur.cuni.cz). Please include your CV and motivation letter.
Preliminary call deadline: 19 April 2026
Note: This is a preliminary call. Shortlisted candidates will be invited to submit a formal PhD application.
Motivation letter (1-2 pages): describe your motivation, research interests, and how your background and skills relate to the selected topic (no generative AI tools, please)
Curriculum Vitae: include education, research experience, MSc thesis (if available), and relevant technical skills

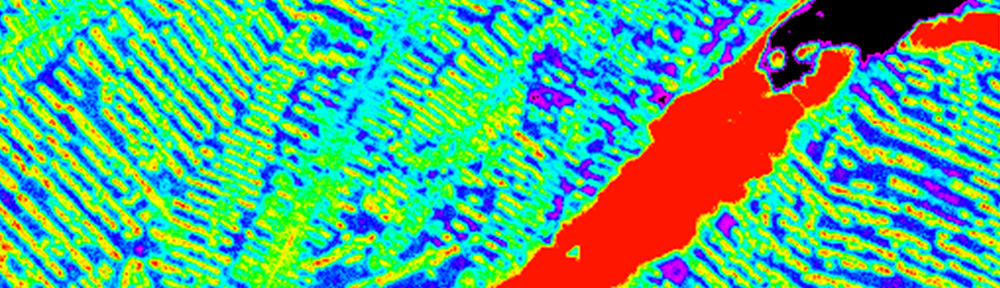



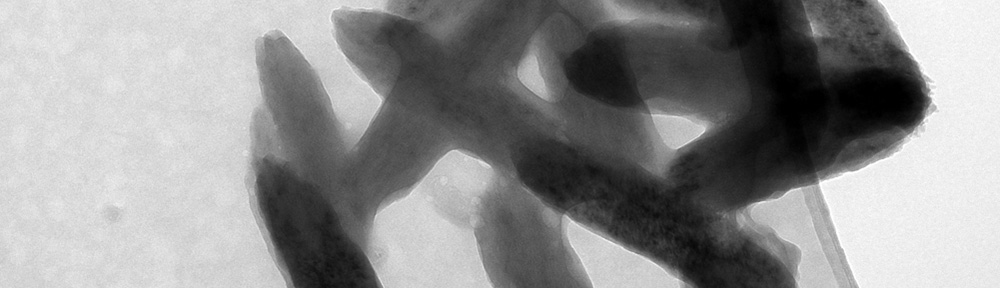


 This study explores the oxidation of arsenopyrite (FeAsS) and löllingite (FeAs2) at high relative humidity (RH: 75%-100%). Long-term oxidation (40 months) experiments show that oxidation of arsenopyrite and löllingite led to formation of different assemblages of secondary phases. Arsenopyrite oxidized to poorly-crystalline ferric arsenate, while löllingite oxidized to scorodite (FeAsO4·2H2O) and arsenolite (As2O3). Our results showed that the exposure of arsenopyrite and löllingite to different RH levels significantly influenced the amounts of newly formed phases. The major environmental impact of sulfide weathering occurs in well aerated environments characterized by high humidity – such as underground workings and some unsaturated mine wastes and tailings deposits.
This study explores the oxidation of arsenopyrite (FeAsS) and löllingite (FeAs2) at high relative humidity (RH: 75%-100%). Long-term oxidation (40 months) experiments show that oxidation of arsenopyrite and löllingite led to formation of different assemblages of secondary phases. Arsenopyrite oxidized to poorly-crystalline ferric arsenate, while löllingite oxidized to scorodite (FeAsO4·2H2O) and arsenolite (As2O3). Our results showed that the exposure of arsenopyrite and löllingite to different RH levels significantly influenced the amounts of newly formed phases. The major environmental impact of sulfide weathering occurs in well aerated environments characterized by high humidity – such as underground workings and some unsaturated mine wastes and tailings deposits. We used an in situ experimental technique with double nylon experimental bags to study the effect of low-cost organic materials (sawdust, wood cubes and hemp shives) on As sulfidation in three naturally As-enriched wetland soils. After 15 months of in situ incubation, all of the organic materials were covered by yellow-black mineral accumulations, dominantly composed of crystalline As4S4 polymorphs (realgar and bonazziite) and reactive Fe(II) sulfides (probably mackinawite). Our findings suggest an authigenic formation of AsS minerals in strongly reducing conditions of experimental bags by a combination of reduced exchange of solutes through the pores of the bag and comparatively fast microbial production of dissolved sulfide. Arsenic sulfide formation, as an effective treatment mechanism for natural and human-constructed wetlands, appears to be favored for As(III)-rich waters with a low Fe(II)/As(III) molar ratio, preventing the consumption of dissolved As and sulfide by their preferential incorporation into natural organic matter, and newly-formed Fe(II) sulfides, respectively.
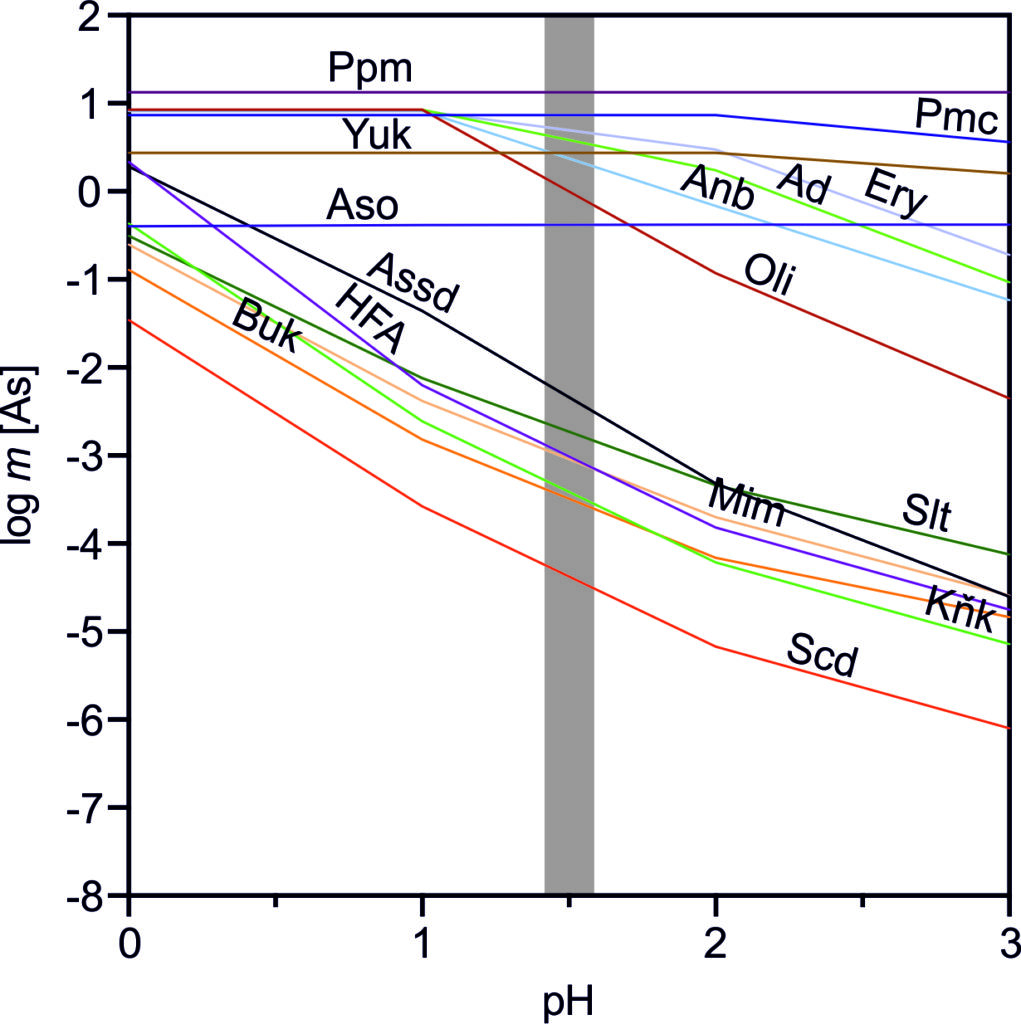
We used an in situ experimental technique with double nylon experimental bags to study the effect of low-cost organic materials (sawdust, wood cubes and hemp shives) on As sulfidation in three naturally As-enriched wetland soils. After 15 months of in situ incubation, all of the organic materials were covered by yellow-black mineral accumulations, dominantly composed of crystalline As4S4 polymorphs (realgar and bonazziite) and reactive Fe(II) sulfides (probably mackinawite). Our findings suggest an authigenic formation of AsS minerals in strongly reducing conditions of experimental bags by a combination of reduced exchange of solutes through the pores of the bag and comparatively fast microbial production of dissolved sulfide. Arsenic sulfide formation, as an effective treatment mechanism for natural and human-constructed wetlands, appears to be favored for As(III)-rich waters with a low Fe(II)/As(III) molar ratio, preventing the consumption of dissolved As and sulfide by their preferential incorporation into natural organic matter, and newly-formed Fe(II) sulfides, respectively. Arsenic-rich stalactites (0.13-294 g/kg As) collected from abandoned adits of the Plavno and Mikulov mines (NW Czech Republic) consist of HFA, HFO, kaňkite, schwertmannite, and hydrous manganese oxide mineral phases. These phases were often combined in one stalactite, forming domains, layers and coprecipitates. The microbial communities indicated autotrophic oxidation of Fe(II), As(III), and reduced sulphur compounds as the most notable biological processes influencing the mineralogy in studied stalactites.
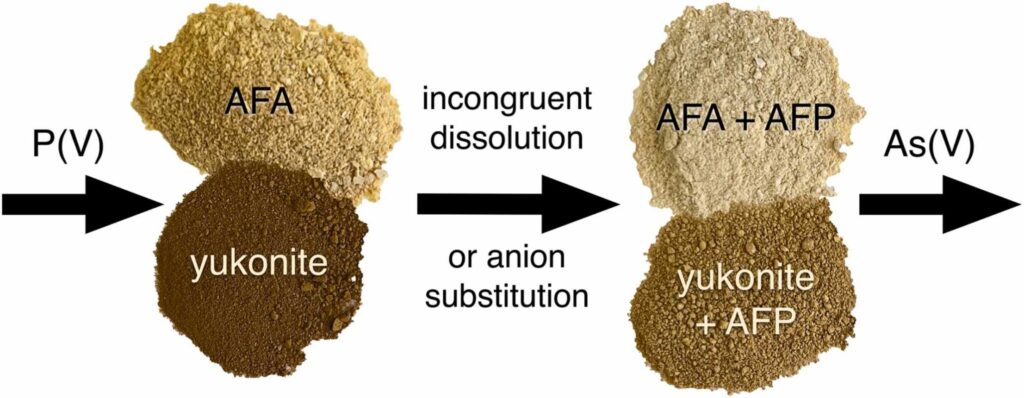
Arsenic-rich stalactites (0.13-294 g/kg As) collected from abandoned adits of the Plavno and Mikulov mines (NW Czech Republic) consist of HFA, HFO, kaňkite, schwertmannite, and hydrous manganese oxide mineral phases. These phases were often combined in one stalactite, forming domains, layers and coprecipitates. The microbial communities indicated autotrophic oxidation of Fe(II), As(III), and reduced sulphur compounds as the most notable biological processes influencing the mineralogy in studied stalactites. This study investigated As-enriched wetland at the Smolotely-Líšnice Au district (Centra Bohemia) using the combination of geochemical (soil and pore water analyses, S isotopes), mineralogical (microprobe, Raman spectrometry) and biological (DNA extraction) method. In this paper, Magda and her co-authors identified a complex assemblage of As and Fe sulfides (realgar, bonazziite, pyrite, greigite) on the fragments of natural organic matter, which play an active role (very fast microbial sulfate reduction vs. slow transfer of solutes) in arsenic immobilization in polluted wetland systems.
This study investigated As-enriched wetland at the Smolotely-Líšnice Au district (Centra Bohemia) using the combination of geochemical (soil and pore water analyses, S isotopes), mineralogical (microprobe, Raman spectrometry) and biological (DNA extraction) method. In this paper, Magda and her co-authors identified a complex assemblage of As and Fe sulfides (realgar, bonazziite, pyrite, greigite) on the fragments of natural organic matter, which play an active role (very fast microbial sulfate reduction vs. slow transfer of solutes) in arsenic immobilization in polluted wetland systems.