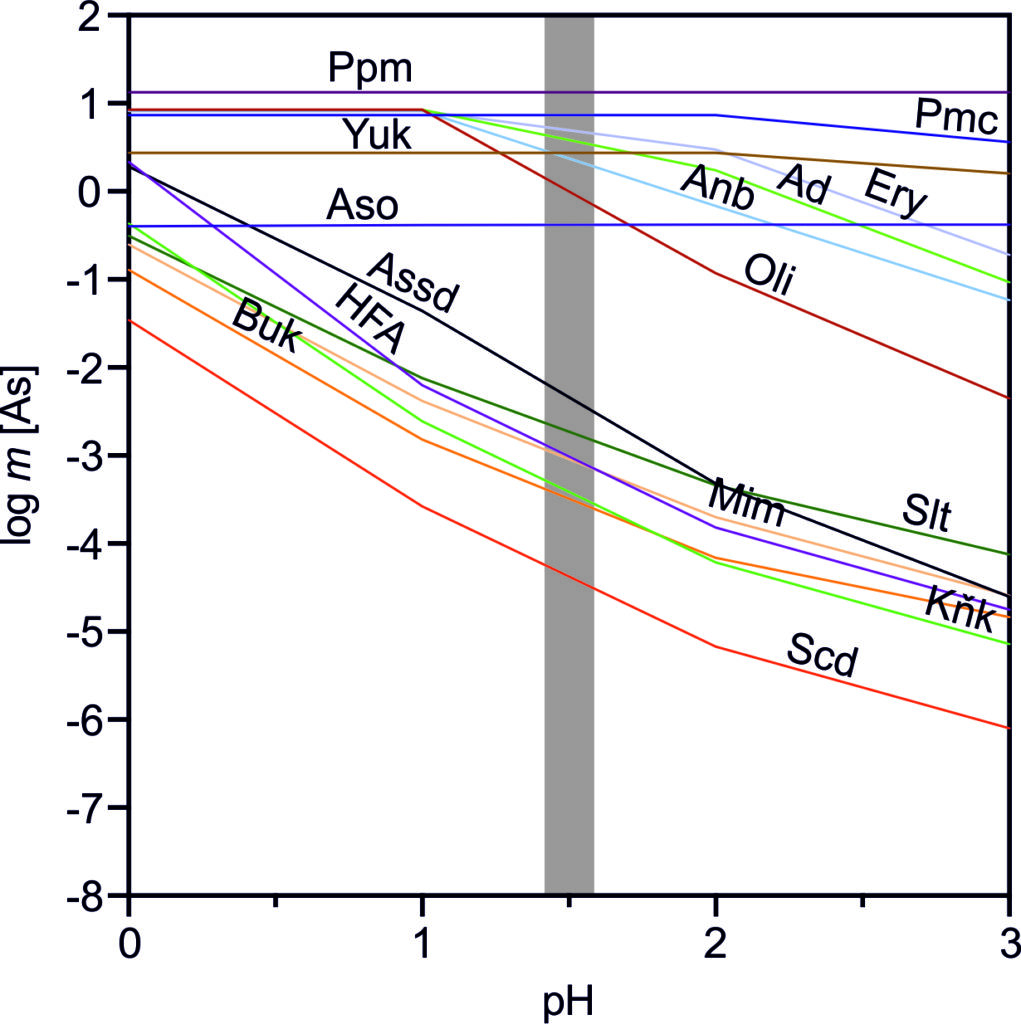
Our new study reveals that the health risk from arsenic-contaminated soil and mine waste is not just about how much arsenic is present—but what form it takes. We tested 16 environmentally important arsenic minerals using a method that mimics human digestion. Results showed bioaccessibility ranged from nearly zero to 100%, depending on the mineral. When mixed with soil, arsenic from low-solubility minerals became even less bioaccessible. These findings highlight the crucial role of mineralogy in assessing arsenic exposure risks.
Drahota P., Ettler V., Košek F. (2025): Arsenic bioaccessibility in environmentally important arsenic minerals. Journal of Hazardous Materials 485, 136838. (DOI)

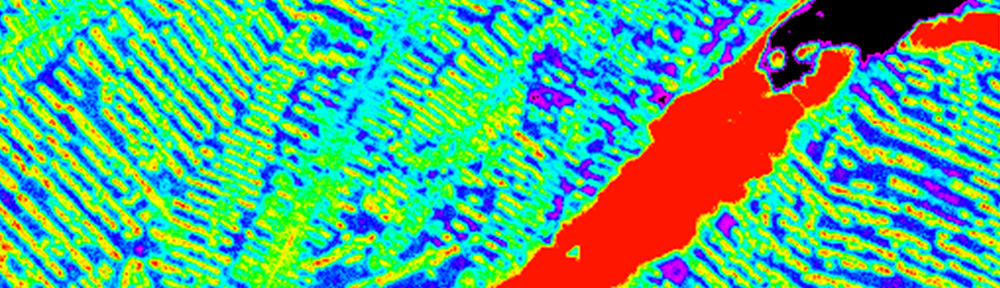



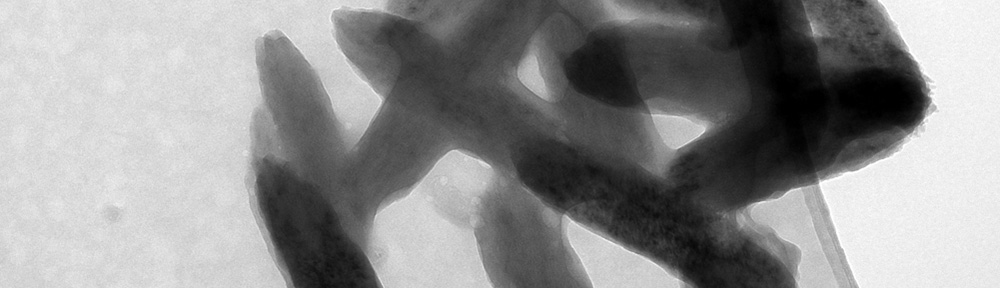


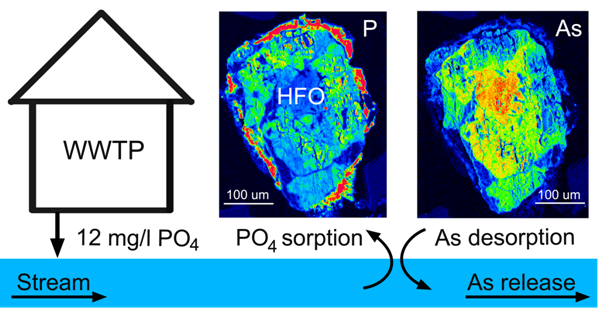
 This study explores the oxidation of arsenopyrite (FeAsS) and löllingite (FeAs2) at high relative humidity (RH: 75%-100%). Long-term oxidation (40 months) experiments show that oxidation of arsenopyrite and löllingite led to formation of different assemblages of secondary phases. Arsenopyrite oxidized to poorly-crystalline ferric arsenate, while löllingite oxidized to scorodite (FeAsO4·2H2O) and arsenolite (As2O3). Our results showed that the exposure of arsenopyrite and löllingite to different RH levels significantly influenced the amounts of newly formed phases. The major environmental impact of sulfide weathering occurs in well aerated environments characterized by high humidity – such as underground workings and some unsaturated mine wastes and tailings deposits.
This study explores the oxidation of arsenopyrite (FeAsS) and löllingite (FeAs2) at high relative humidity (RH: 75%-100%). Long-term oxidation (40 months) experiments show that oxidation of arsenopyrite and löllingite led to formation of different assemblages of secondary phases. Arsenopyrite oxidized to poorly-crystalline ferric arsenate, while löllingite oxidized to scorodite (FeAsO4·2H2O) and arsenolite (As2O3). Our results showed that the exposure of arsenopyrite and löllingite to different RH levels significantly influenced the amounts of newly formed phases. The major environmental impact of sulfide weathering occurs in well aerated environments characterized by high humidity – such as underground workings and some unsaturated mine wastes and tailings deposits.
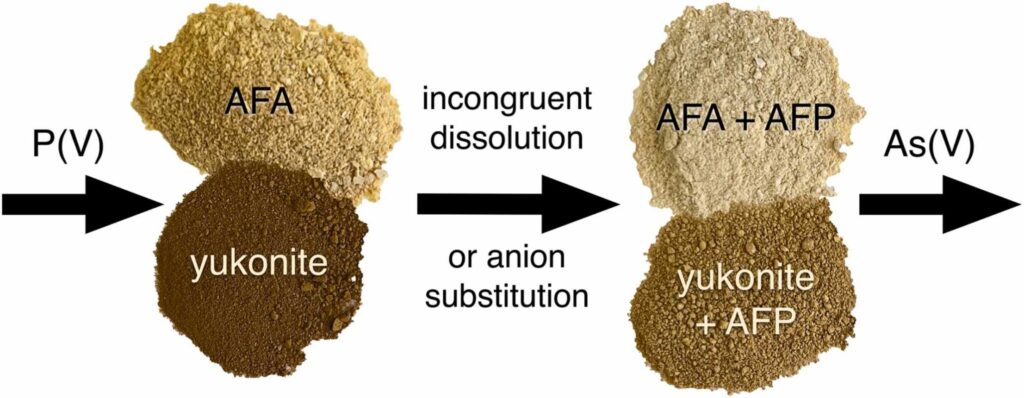
 We used an in situ experimental technique with double nylon experimental bags to study the effect of low-cost organic materials (sawdust, wood cubes and hemp shives) on As sulfidation in three naturally As-enriched wetland soils. After 15 months of in situ incubation, all of the organic materials were covered by yellow-black mineral accumulations, dominantly composed of crystalline As4S4 polymorphs (realgar and bonazziite) and reactive Fe(II) sulfides (probably mackinawite). Our findings suggest an authigenic formation of AsS minerals in strongly reducing conditions of experimental bags by a combination of reduced exchange of solutes through the pores of the bag and comparatively fast microbial production of dissolved sulfide. Arsenic sulfide formation, as an effective treatment mechanism for natural and human-constructed wetlands, appears to be favored for As(III)-rich waters with a low Fe(II)/As(III) molar ratio, preventing the consumption of dissolved As and sulfide by their preferential incorporation into natural organic matter, and newly-formed Fe(II) sulfides, respectively.
We used an in situ experimental technique with double nylon experimental bags to study the effect of low-cost organic materials (sawdust, wood cubes and hemp shives) on As sulfidation in three naturally As-enriched wetland soils. After 15 months of in situ incubation, all of the organic materials were covered by yellow-black mineral accumulations, dominantly composed of crystalline As4S4 polymorphs (realgar and bonazziite) and reactive Fe(II) sulfides (probably mackinawite). Our findings suggest an authigenic formation of AsS minerals in strongly reducing conditions of experimental bags by a combination of reduced exchange of solutes through the pores of the bag and comparatively fast microbial production of dissolved sulfide. Arsenic sulfide formation, as an effective treatment mechanism for natural and human-constructed wetlands, appears to be favored for As(III)-rich waters with a low Fe(II)/As(III) molar ratio, preventing the consumption of dissolved As and sulfide by their preferential incorporation into natural organic matter, and newly-formed Fe(II) sulfides, respectively.
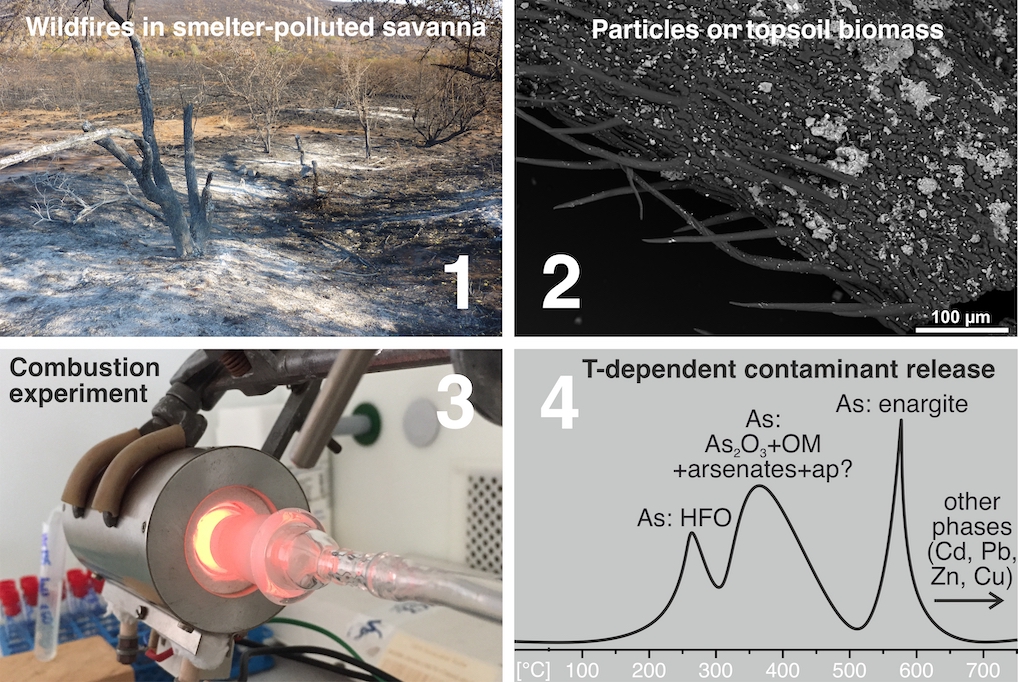 The temperature-dependent releases of metal(loid)s (As, Cd, Cu, Pb, Zn) from biomass-rich savanna soils collected near a Cu smelter in Namibia have been studied under simulated wildfire conditions. For this purpose, new wildfire-simulating setups were introduced. Laboratory single-step combustion experiments (250–850 °C) and experiments with a continuous temperature increase and online ICP-OES detection (25–750 °C) were coupled with mineralogical investigations of the soils, ashes, and aerosols. The results indicate that metals are dominantly concentrating in the ash residue, and part of As is remobilized depending on temperature. Therefore, the active and abandoned mining and smelting sites, especially those highly enriched in As, should be protected against wildfires, which can be responsible for substantial As re-emissions.
The temperature-dependent releases of metal(loid)s (As, Cd, Cu, Pb, Zn) from biomass-rich savanna soils collected near a Cu smelter in Namibia have been studied under simulated wildfire conditions. For this purpose, new wildfire-simulating setups were introduced. Laboratory single-step combustion experiments (250–850 °C) and experiments with a continuous temperature increase and online ICP-OES detection (25–750 °C) were coupled with mineralogical investigations of the soils, ashes, and aerosols. The results indicate that metals are dominantly concentrating in the ash residue, and part of As is remobilized depending on temperature. Therefore, the active and abandoned mining and smelting sites, especially those highly enriched in As, should be protected against wildfires, which can be responsible for substantial As re-emissions.